I have been following the news on Covid-19 with many of you and have had the opportunity to participate in webinars from IFM and others in the health care community. As communities start considering going back to work, accurate and accessible testing will be imperative. I’m a Gynecologist, not a Covid expert, but these are extreme times. I wanted to summarize information from reliable sources for my community so that folks had a framework to understand the timing, accuracy, and clinical implications of testing.
What are symptoms of Covid-19?
The CDC recently updated its symptoms for COVID-19.
People with COVID-19 have had a wide range of symptoms reported – ranging from mild symptoms to severe illness.
- Fever
- Cough
- Shortness of breath or difficulty breathing
- Chills
- Repeated shaking with chills
- Muscle pain
- Headache
- Sore throat
- New loss of taste or smell
When will symptoms occur after an exposure?
Time of exposure to symptom development ranges from 2-21 days.
- >90% of people who develop symptoms will do so within 14 days.
- Up to 10% of people may develop symptoms later than 14 days.
- Many people never develop symptoms but may still develop antibody responses. The timing and level of antibody responses in asymptomatic people is presently unknown.
How long will I be infectious?
Viral shedding can begin 3-21 days after exposure (i.e., preceding symptoms).
- Viral RNA shedding can occur in nasopharynx, oropharynx, saliva, and sputum for up to 21 days after symptom resolution.
- Testing evaluates viral RNA, not shedding of intact virus (i.e., infectivity).
- It appears that intact viral shedding is complete 14 days after symptoms begin, though viral RNA shedding continues (as noted above).
Types of testing
First of all, there are 2 main types of testing. There is a PCR test which is usually done from a nasopharyngeal swab, sputum, saliva and this test is looking for viral RNA. It’s checking to see if the patient is acutely infected (are they sick now). This is usually done in a patient that is symptomatic with a fever, cough, headache, chills, acute loss of smell or has a known exposure. (The last part really gets me. Our ability to provide adequate testing has been so limited, how are folks supposed to know if they have been exposed or not?) One would think that if the test were testing the actual RNA of the virus that it would be pretty accurate, but unfortunately that isn’t the case. RT- PCR is estimated to have up to a 30% false negative rate. This means that if your Covid-19 PCR says that you do not have the disease, there is a roughly 1 in 3 chance that you do!
Reasons why a PCR test could be a false negative:
- Inappropriate collection site – Nasopharyngeal swabs are better at detecting than throat swabs.
- Inadequate amount of sample taken – have you ever pulled away when someone was trying to collect a nasopharyngeal swab – it’s uncomfortable.
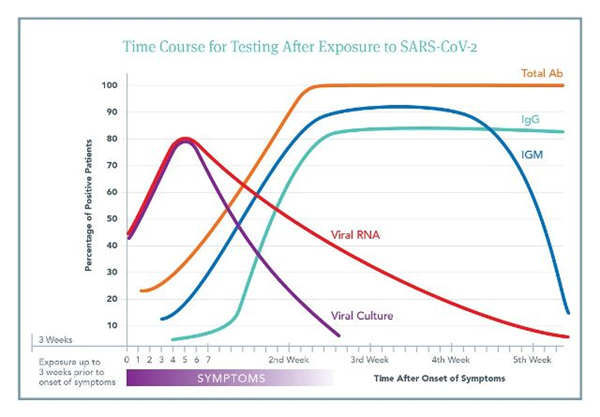
- Timing of collection – the diagnostic window is 3-21 days. As you can see in the graph below, viral RNA peaks around day 4 and starts to decline after then.
- Handling, transport and storage of swabs as well as the presence of interfering substances or contamination can also contribute to false negatives.
IGG /IGM testing
The second type of testing is checking for immune response. If there is an immune response, the body has encountered the virus. The body’s immune system has different branches. IgA antibodies are usually on mucosal surfaces, IGM antibodies are the early responders while IGG antibodies are present with long term immunity. (The way I used to remember this was that IGM is iMmediate and IGG is Gone.) Typically, when folks have IGG antibodies, we think of them having immunity. IGG/ IGM testing can be done with whole blood, plasma, serum or finger stick testing.
Aren’t there a lot of questionable test companies popping up? How do I know who to trust?
The NYT published last week concerning reports on how few testing companies were FDA approved (only 4) and results of questionable accuracy. Without a degree in microbiology how do you determine which test is reliable? IFM lists several questions to ask a testing company. I have used KMBO for food sensitivity testing and have been impressed with how their company has responded to answering the following questions:
- What are the control samples? Is the lab using clinical samples as opposed to ‘spiked’ (i.e., artificial) samples? Yes, KMBO test has been validated with clinical samples in over 2 million patients in China.
- Did the positive test population have symptoms in addition to a positive PCR test? Yes
- Did the assay evaluate samples that had exposure to other respiratory viruses such as influenza and RSV, as well as other coronaviruses, to determine cross reactivity and specificity? Yes
- KMBO IGG/IGM testing reported a sensitivity (the percentage of truly infected individuals who will test positive on a test) of 87.3% (95% CI 80.40%-92%) and a specificity (percentage of truly uninfected individuals who test negative) of 100%
- Note: KMBO is a CLIA approved lab in the USA. Their test is approved by the Chinese FDA. They have applied to get FDA approval in the USA, but this is still pending.
Isn’t the virus mutating? How do I know if the test is effective?
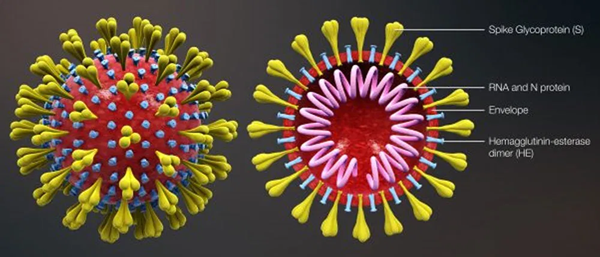
To answer the question, first we need to see the components of the virus:
Currently there are 6 identified strains of COVID-19. If the virus keeps changing, how do we know that what we are testing is correct? The KMBO test kit is measuring antibodies to Nuclear protein, labeled N protein above. This protein appears to be more stable than the S and M proteins. The other thing to consider is that there are lots of corona viruses out there— we want to make sure that the test is measuring COVID-19 and not measuring exposure to SARS or another virus. The N Protein in the KMBO test won’t cross react with other coronaviruses.
When should IGM/ IGG testing be done?
At least 15 days after fever:
- Testing performed in the first week after symptoms yields a positive antibody result only 38% of the time.
- 15-18 days after fever 90% will have IGG+ results.
- The CDC recommends testing 28 days after fever at which point 100% will have +IGG.
If you have + IGG immunity is that a healthy “passport”?
While we use IGG testing in lots of areas of medicine, we are still learning about this virus. I caution against folks with positive IGG antibodies to Covid-19 thinking that they are immune and engaging in behavior that puts others at risk. The WHO’s position on this is listed here:
At this point in the pandemic, there is not enough evidence about the effectiveness of antibody-mediated immunity to guarantee the accuracy of an “immunity passport” or “risk-free certificate.” People who assume that they are immune to a second infection because they have received a positive test result may ignore public health advice. The use of such certificates may therefore increase the risks of continued transmission.
WHO, “Immunity passports” in the context of COVID-19
Should I get tested?
I’m offering KMBO IGG/ IGM testing to my patients because many have been ill but not ill enough to require acute care services. They haven’t been tested and it would give them peace of mind to know if they have some immunity moving forward. I do not suggest that if someone tests positive for IGG antibodies that they are “100% immune.” We don’t have data to prove this, nor do we have data to suggest that IGG+ folks are not shedding virus that could be passed to others.
How much does testing cost?
The test from KMBO is $150 and this price includes overnight shipping to get the kit to you and to ship the kit back to the lab. It requires you to do a fingerstick at home and provide 3 drops of blood on filter paper. The results will go to Dr. Watson who will charge a $50 administrative fee to call you and review your results for 10 minutes.
Can you test my family members?
No. Legally, we can only test those who are patients of record at Hormone Wellness MD. Patients who saw me years ago at Boylan are not candidates unless they have seen me at my new practice.
How do I get the kit?
Go to this website to order the kit:
https://kbmodiagnostics.com/covid-19-test-kit-order-form-patients/
Message us through the portal or call our office for the code you will need. Once we confirm you are a patient of record, we will give you the code and schedule you for a 10-minute phone appointment with Dr. Watson.